MULTI-seq
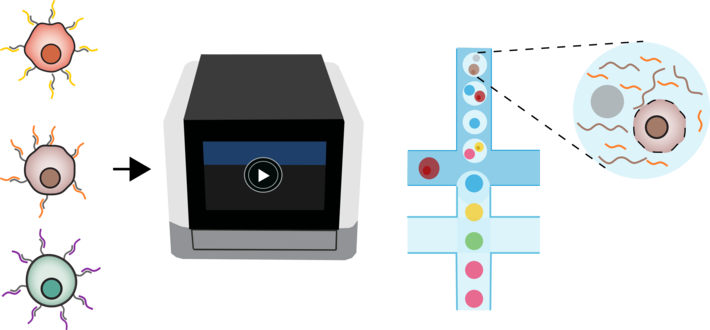
After years of innovations that led to improvements in sensitivity and throughput of single cell RNA sequencing methods, the 10x Genomics Chromium platform has emerged as one of the most widely used single cell RNA seq technology. Two recent benchmarking studies confirmed its remarkable performance when assaying tens of thousands of cells in a single run (Ding et al. 2019; Mereu et al. 2019). This is enabled through miniaturization and single cell isolation using microfluidic devices as initially introduced by Drop-seq and InDrops. However, one big caveat that limits 10x’s usability beyond cell atlas projects is that sample multiplexing is not easily possible, especially when complex experimental designs with multiple replicates and perturbations are performed. A number of techniques have been developed to overcome this limitation and incorporate additional biologically relevant readouts, like protein expression, CRISPR editing and more (ExCITEseq; Perturb-seq). MULTI-seq, a particularly promising method, uses lipid tagged barcode oligos to label cells prior to processing using 10x Genomics. The major advantages of this method are the easy and inexpensive workflow and the broad applicability on virtually all mammalian cells as well as nuclei.
Links: https://github.com/chris-mcginnis-ucsf/MULTI-seq
Paper link: https://www.nature.com/articles/s41592-019-0433-8